|
It states that the University is guided by the ethical principles of the Belmont Report, and will comply with federal regulations (45 Code of Federal Regulations Part 46, or simply 45 CFR 46) (PDF) for all federally-funded human subjects research. An FWA is a binding written agreement between the University and DHHS. Revision 2 approved by Assistant VP Regulatory Affairs on 11-24-2015. Revision 3 approved by Assistant VP Regulatory Affairs on 11-27-2021.NYUAD has filed an assurance of compliance, called a Federalwide Assurance (FWA) (PDF), with the Office for Human Research Protections (OHRP) in the US Department of Health and Human Services (DHHS). Revision 1 approved by the VP Research & Graduate Studies on 7-21-2011. This policy and procedure supersedes those previously drafted.Īpproved By: Vice President of Research and Graduate Studies, 3-3-2005. Vulnerable classes of subjects should be given special protection and not be unduly selected as research subjects due to their ready availability or dependent status.įull text of The Belmont Report is available at Selection of individuals or classes of individuals should be fair. Justice: The risks of research should be equally distributed and should not unduly involve persons from groups unlikely to be among the beneficiaries of the research. In some cases where research may pose risk to individual subjects with no direct benefit to them the principle of beneficence requires careful assessment of the benefits to others or to society. 
One group entitled to special protections is prisoners who may be subtly coerced or unduly influenced.īeneficence: Researchers are obligated to do no harm by maximizing possible benefits and reducing possible risks to subjects. Special protection should be given to individuals with diminished autonomy and/or of special circumstances since they may not be able to make a considered judgment even if they are adequately informed. They should voluntarily enter into research by being adequately informed. Respect for Persons: Individuals should be treated as autonomous agents. The principles found in the Belmont Report are summarized as follows: Michigan State University (MSU) applies the ethical principles found in the Belmont Report to all of its activities related to human subject research, regardless of funding. The Belmont Report was produced by the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research and became the foundation of U.S. Refusal of patient to participate in research must never interfere with physician-patient relationshipįull text of the Declaration of Helsinki is available at The Belmont Report - 1979.Well-being of subject takes precedence over interests of science and society.Health of patient is first consideration.The Declaration of Helsinki includes principles such as: 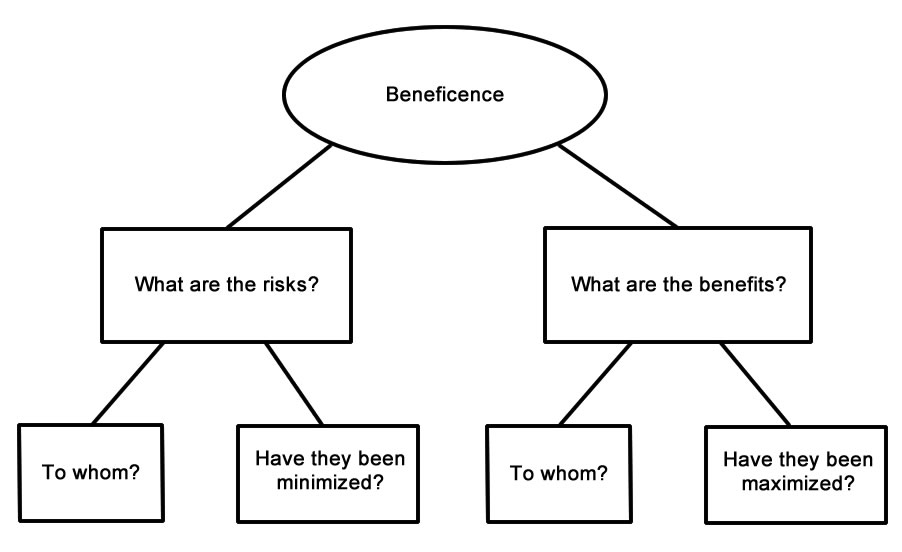
It is a statement of ethical principles for medical human subject research and discusses research versus clinical care. The Declaration of Helsinki was adopted by the World Medical Assembly (WMA) in Helsinki, Finland in 1964 and is periodically amended by the WMA. The Nuremberg Code includes principles such as:įull text of The Nuremberg Code is available at The Declaration of Helsinki - 1964 The Nuremberg Code was adopted by the United Nations General Assembly and is considered international “common law.” The Nuremberg Code was developed out of the trials of Nazi war criminals, including those involved in medical experiments. 
For more information on ICH GCP (E6) (R2), see the Human Research Protection Program (HRPP) Manual 2-4 “International Conference on Harmonization Good Clinical Practice (E6) (R2).” The Nuremberg Code - 1949 The ICH GCP (E6) (R2) provides international guidelines for the conduct of clinical trials. 
In addition, clinical human subject research may also follow the International Conference on Harmonisation – Guidelines for Good Clinical Practice (ICH GCP) (E6) (R2). Short overviews of these principles are provided below. Ethical principles set forth in these founding documents should be upheld during the conduct, review, and oversight of research by investigators, research coordinators, key personnel, employees, students, organizational officials, IRB members, and IRB staff. The contemporary protections for human subjects of research are based on three founding documents: the Nuremberg Code (1949), the Declaration of Helsinki (1964), and the Belmont Report (1979). MSU HRPP Manual Section 2-1 Ethical Principles
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
